 BS Research's biocompatible surface silicone gel-filled breast implant is a smooth type that rarely related to the BIA-ALCL and it is developed to have a biocompatible surface using BS research's PMPC grafting technology.
BS Research's biocompatible surface silicone gel-filled breast implant is a smooth type that rarely related to the BIA-ALCL and it is developed to have a biocompatible surface using BS research's PMPC grafting technology.
: The company owns a patented method for producing a new type of shell that compensates for the disadvantages of silicone while maintaining the original properties of silicone and PMPC. It can reduce various adverse reactions associated with the implantation of silicone gel-filled breast implants and is also expected to contribute reducing 'capsular contracture,' the biggest reason of breast implant removal and replacement surgery.
It can reduce various adverse reactions associated with the implantation of silicone gel-filled breast implants and is also expected to contribute reducing 'capsular contracture,' the biggest reason of breast implant removal and replacement surgery.
Silicone gel-filled breast implant with physical properties
 When the surface of a silicone gel-filled breast implant is modified into a biocompatible surface, property degradation inevitably occurs due to changes in the surface structure. However, BS Research can minimize the degradation and secure physical properties that meet the requirements.
When the surface of a silicone gel-filled breast implant is modified into a biocompatible surface, property degradation inevitably occurs due to changes in the surface structure. However, BS Research can minimize the degradation and secure physical properties that meet the requirements.
Published paper
 BS Research's joint research team led by SMG-SNU Boramae Hospital Professor Ji-Ung Park and Seoul National University Professor Yan Lee presented results on the 'Efficient reduction of fibrous capsule formation around silicone gel-filled breast implants densely grafted with 2-methacryloyloxyethyl phosphorylcholine (MPC) polymers by heat-induced polymerization.'
BS Research's joint research team led by SMG-SNU Boramae Hospital Professor Ji-Ung Park and Seoul National University Professor Yan Lee presented results on the 'Efficient reduction of fibrous capsule formation around silicone gel-filled breast implants densely grafted with 2-methacryloyloxyethyl phosphorylcholine (MPC) polymers by heat-induced polymerization.'
: Results were published as a cover paper in the March 2020 issue of an international journal from the Royal Society of Chemistry, the world-leading chemistry community.
Efficacy of BMH™ Shell Silicone Gel-filled Breast Implant
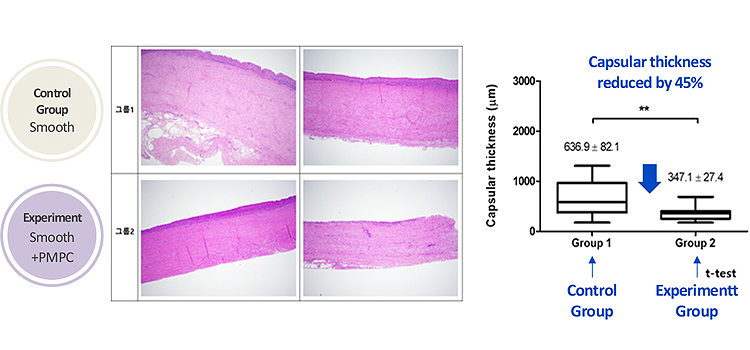
 In a study that measured the capsular thickness of non-grafted and grafted silicone gel-filled breast implants 24 weeks after being inserted to pigs, results showed that the capsular thickness of the two were as follows:
In a study that measured the capsular thickness of non-grafted and grafted silicone gel-filled breast implants 24 weeks after being inserted to pigs, results showed that the capsular thickness of the two were as follows:
- Grafted breast implants showed significant 45% less capsular thickness
- The non-thickening capsular can serve as evidence of its capsular contracture prevention effect
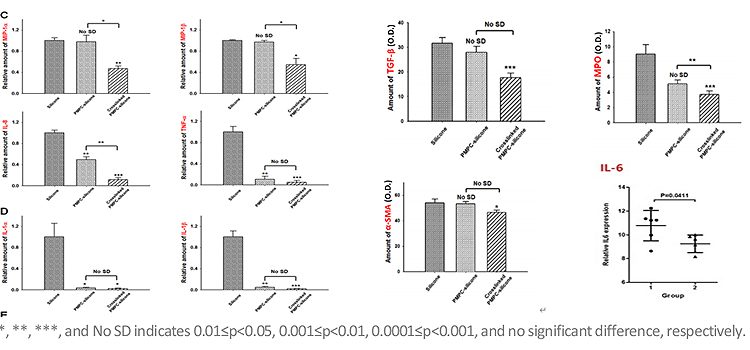
 The lower cytochemical level in the grafted silicone gel-filled breast implant when compared with the non-grafted implant indicate that:
The lower cytochemical level in the grafted silicone gel-filled breast implant when compared with the non-grafted implant indicate that:
- The biomarkers related to foreign body reaction and inflammatory response were significantly lower with the grafted implant
- Expression of inflammatory cells and collagen was significantly lower
- These can serve as evidence that the grafted silicone gel breast implant prevents excessive foreign body reaction and inflammation
| Differences in Capsular Thickness According to Grafting Status |
|---|
 |
| Differences in Inflammatory Markers According to Grafting Status |
|---|
 |
Biological Safety
 BS Research performed a biocompatibility tests(Cytotoxicity, Intracutaneous Reactivity, Skin Sensitization, Acute Systemic Toxicity, Material-mediated Pyrogenicity, Genotoxicity, Rat-implantation, Rat-Subchronic Toxicity tests) according to ISO10993 at the GLP-certified testing lab.
BS Research performed a biocompatibility tests(Cytotoxicity, Intracutaneous Reactivity, Skin Sensitization, Acute Systemic Toxicity, Material-mediated Pyrogenicity, Genotoxicity, Rat-implantation, Rat-Subchronic Toxicity tests) according to ISO10993 at the GLP-certified testing lab. BS Research's biocompatible surface silicone gel-filled breast implant is the only product that has biocompatibility and a BMH™Shell, which has similar properties to the cell membranes
BS Research's biocompatible surface silicone gel-filled breast implant is the only product that has biocompatibility and a BMH™Shell, which has similar properties to the cell membranes
- PMPC is the material that has been used and approved for the medical devices such as coronary artery stents and artificial joints due to its benefit of biocompatibility.
- BS Research grafted PMPC on the shell of silicone gel-filled breast implant for the first time, and the grafting technology provides a differentiated effectiveness, semi-permanent biocompatibility.
Performance & Physicochemical Properties
 BS Research completed development of silicone gel-filled breast implant with BMH™ Shell and it meets the regulatory authorities' global standard requirements.
BS Research completed development of silicone gel-filled breast implant with BMH™ Shell and it meets the regulatory authorities' global standard requirements.
Current status of breast implant
 Completed biological safety tests
Completed biological safety tests Completed performance and physicochemical property tests
Completed performance and physicochemical property tests
Plans for Clinical Trials in Korea and Abroad
 Clinical trial in Korea: Planned IDE/IRB approval in 2023
Clinical trial in Korea: Planned IDE/IRB approval in 2023 Clinical trials in the U.S. and Europe: Planning to promote clinical trials, including overseas partnership or license-out
Clinical trials in the U.S. and Europe: Planning to promote clinical trials, including overseas partnership or license-out
Plans for Overseas Market Expansion
 Considering and reviewing overseas partnerships and licensing-out options
Considering and reviewing overseas partnerships and licensing-out options
: Technology transfer; establishment of joint venture; supplying the company's biocompatible surface construction technology based on grafting technology, etc.
